투석환자분에게 팍스로비드를 사용할 수 있을까? 그 전에는 안전성에 대한 데이터가 부족하여 투석환자분께 드릴 수 없었으나, 투석환자도 사용가능하다는 여러 후향적 연구 결과들이 나오고 있어 관련 자료들을 소개합니다.
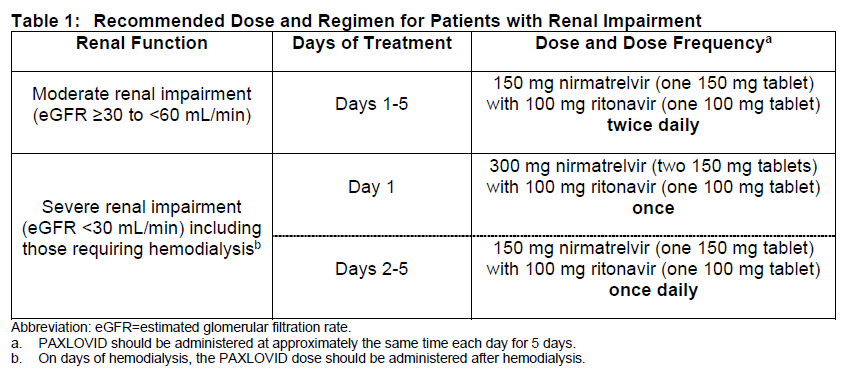
팍스로비드 신기능에 따른 용량 조절
Medscape – nirmatrelvir/ritonavir (Rx)
- Mild (eGFR ≥60 to <90 mL/min): No dosage adjustment required
- Moderate (eGFR ≥30 to <60 mL/min): Decrease to 150 mg nirmatrelvir plus 100 mg ritonavir BID x 5 days
- Severe (eGFR <30 mL/min) including hemodialysis
- Day 1: 300 mg nirmatrelvir plus 100 mg ritonavir PO once
- Days 2-5: 150 mg nirmatrelvir plus 100 mg ritonavir PO once daily
- On days patients with severe renal impairment undergo hemodialysis, the dose should be administered after hemodialysis
Paxlovid Fact sheet (Revised EUA Authorized Date: 02/2025)
2.1 Important Dosage and Administration Information for Emergency Use of PAXLOVID
PAXLOVID is nirmatrelvir tablets co-packaged with ritonavir tablets. There are two different dose packs available:
- PAXLOVID (nirmatrelvir; ritonavir) co-packaged for oral use 300 mg;100 mg [see Dosage and Administration (2.2)].
- PAXLOVID (nirmatrelvir; ritonavir) co-packaged for oral use 150 mg;100 mg for patients with moderate renal impairment [see Dosage and Administration (2.3)].
- PAXLOVID (nirmatrelvir; ritonavir) co-packaged for oral use 300 mg;100 mg (Day 1) and 150 mg;100 mg (Days 2-5) for patients with severe renal impairment [see Dosage and Administration (2.3)].
Nirmatrelvir must be co-administered with ritonavir. Failure to correctly co-administer nirmatrelvir with ritonavir may result in plasma levels of nirmatrelvir that are insufficient to achieve the desired therapeutic effect.
PAXLOVID is contraindicated with drugs that are primarily metabolized by CYP3A and for which elevated concentrations are associated with serious and/or life-threatening reactions and drugs that are strong CYP3A inducers where significantly reduced nirmatrelvir or ritonavir plasma concentrations may be associated with the potential for loss of virologic response and possible resistance. There are certain other drugs for which concomitant use with PAXLOVID should be avoided and/or dose adjustment, interruption, or therapeutic monitoring is recommended. Drugs listed in this section are a guide and not considered a comprehensive list of all drugs that may be contraindicated with PAXLOVID. The healthcare provider should consult other appropriate resources such as the prescribing information for the interacting drug for comprehensive information on dosing or monitoring with concomitant use of a strong CYP3A inhibitor like PAXLOVID [see Drug Interactions (7.3)]:
Drugs that are primarily metabolized by CYP3A for which elevated concentrations are associated with serious and/or life-threatening reactions [see Drug Interactions (7.3)]:
- Alpha 1-adrenoreceptor antagonist: alfuzosin
- Antianginal: ranolazine
- Antiarrhythmic: amiodarone, dronedarone, flecainide, propafenone, quinidine
- Anti-gout: colchicine (in patients with renal and/or hepatic impairment [see Table 2, Drug Interactions (7.3)])
- Antipsychotics: lurasidone, pimozide
- Benign prostatic hyperplasia agents: silodosin
- Cardiovascular agents: eplerenone, ivabradine
- Ergot derivatives: dihydroergotamine, ergotamine, methylergonovine
- HMG-CoA reductase inhibitors: lovastatin, simvastatin (these drugs can be temporarily discontinued to allow PAXLOVID use [see Table 2, Drug Interactions (7.3)])
- Immunosuppressants: voclosporin
- Microsomal triglyceride transfer protein inhibitor: lomitapide
- Migraine medications: eletriptan, ubrogepant
- Mineralocorticoid receptor antagonists: finerenone
- Opioid antagonists: naloxegol
- PDE5 inhibitor: sildenafil (Revatio®) when used for pulmonary arterial hypertension (PAH)
- Sedative/hypnotics: triazolam, oral midazolam
- Serotonin receptor 1A agonist/serotonin receptor 2A antagonist: flibanserin
- Vasopressin receptor antagonists: tolvaptan
University of illinois chicago : What evidence evaluates the use of Paxlovid™ (nirmatrelvir/ritonavir) in severe renal impairment?
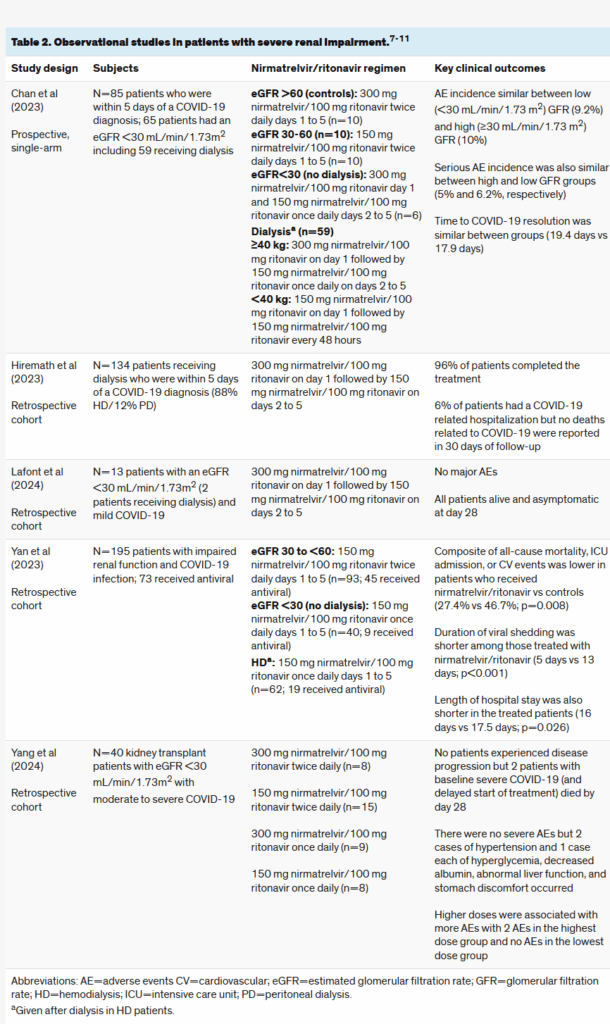
표 2에 포함된 연구들은 경증, 중등도, 또는 중증 신장애 환자를 모두 포함하는 환자군을 대상으로 하였다.중증 신부전(eGFR <30 mL/min/1.73 m²) 환자에서 닌마트렐비르/리토나비르 사용을 뒷받침하는 현재 문헌은 주로 후향적 코호트 연구에 기반하고 있다. 전반적으로, 닌마트렐비르/리토나비르는 이러한 환자에게 1일차 닌마트렐비르 300 mg/리토나비르 100 mg, 이후 2~5일차 닌마트렐비르 150 mg/리토나비르 100 mg 1일 1회 투여가 안전하고 효과적이라는 점을 지지한다. 투석 환자의 경우 체중이 40kg 이상인 경우 동일한 용량이 적절해 보이며, 체중이 더 적은 환자에서는 저용량이 적절하다. 중등도 신부전 환자에 대해서는, 이러한 관찰 연구에서 사용된 용량이 제품 설명서와 대체로 일치한다.
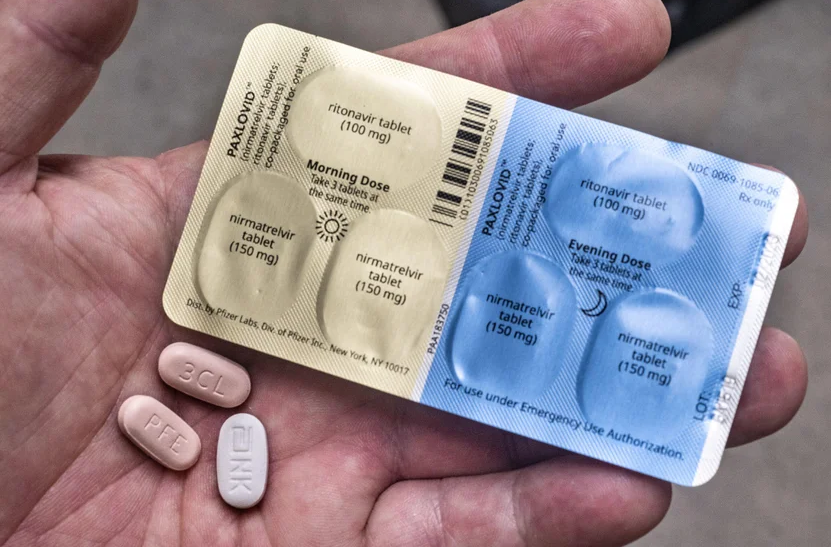
정리 : 투석환자 팍스로비드 용량
- 팍스로비드는 [nirmatrelvir 150mg 2T + ritonavir 100mg 1T] 씩 총 3T가 한 묶음로 포장
- 1일차 [nirmatrelvir 150mg 2T + ritonavir 100mg 1T] 1일 1회
- 2~5일차 [nirmatrelvir 150mg 1T + ritonavir 100mg 1T] 1일 1회
아직은 투석환자분께 팍스로비드를 저용량으로 사용하는 것은 논란의 여지가 있는 것 같습니다. 하지만 후향적 연구에서도 아직 큰 문제는 없었으며, 여러 문헌에도 등장하는 것을 보면 하나의 치료 옵션으로 기억해두시면 좋을 것 같습니다.