Primary hyperparathyroidism
Nature reviews endocrinology
Vol 14, 2018. 2
Epidemiology and pathogenesis
PHPT is caused by a solitary parathyroid adenoma in 80% of cases, whereas four-gland hyperplasia accounts for 10–15%, multiple denomas for 5% and parathyroid cancer for <1% of cases. Incidence estimates for PHPT vary from ~0.4 to 82 cases per 100,000. The incidence of PHPT increases with age and is higher in women and African Americans than in men and other racial groups, respectively. Half of all patients with PHPT are postmenopausal women, although the disorder can occur at any age.
The underlying cause of sporadic PHPT is unknown in most cases. Ionizing radiation, especially in childhood, is a risk factor. Chronic lithium use, which decreases the sensitivity of the parathyroid glands to calcium, is also associated with the development of PHPT. Two such genes documented as contributing to the development of PHPT are CCND1 (which encodes cyclin D1) and MEN1 (which encodes menin). Somatic mutations in MEN1 occur in 12–35% of sporadic adenomas, whereas rearrangement or overexpression of CCND1 can occur in 20–40%. In inherited or familial forms of PHPT, which represent about 5–10% of cases.
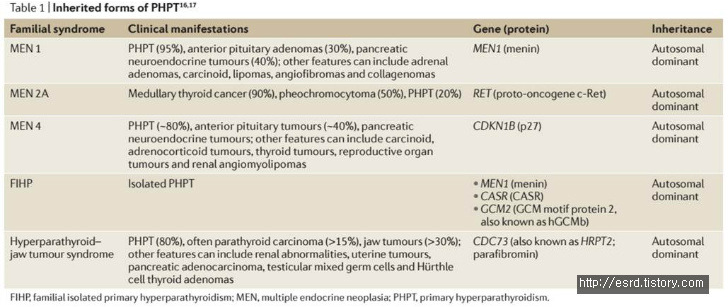
The tumour suppressor MEN1 in multiple endocrine neoplasia type 1 syndrome and familial isolated primary hyperparathyroidism(FIHP) the proto-oncogene RET in MEN 2A Syndrome CDKN1B in MEN 4 syndrome; inactivating mutations in CASR (which encodes the calcium-sensing receptor) in FIHP GCM2 in FIHP and CDC73 in hyperparathyroidism–jaw tumour syndrome, which is also associated with an increased risk of parathyroid carcinoma. Mutations in PRUNE2 (which encodes protein prune homologue 2) have also been associated with the development of parathyroid cancer microRNA 296 might be a novel tumour-suppressor gene in parathyroid carcinoma. The genetics and characteristics of familial hypocalciuric hypercalcaemia (FHH), which is not considered a form of PHPT.
Pathophysiology and (differential) diagnosis
Loss of normal feedback, suppression of serum levels of calcium upon the synthesis and secretion of PTH, due to increased parathyroid cell mass and/or a reduction in the number of CASR proteins on parathyroid cells. As a result, increased levels of calcium are needed to suppress PTH levels. The diagnosis of PHPT is established biochemically and can be confirmed by documenting hypercalcaemia with a simultaneously elevated intact PTH level. Non-parathyroid causes of hypercalcaemia (such as malignancy or granulomatous disease) are associated with suppressed levels of PTH.
Parathyroid imaging has no role in the diagnosis of PHPT. Imaging studies assist the parathyroid surgeon in identifying the anatomic position of abnormal gland(s) when planning parathyroidectomy. Negative imaging, which is frequent in multi-glandular PHPT, is not inconsistent with the diagnosis of PHPT. False-positive tests occur often in those with concurrent nodular thyroid disease.
To differentiate PHPT from FHH, which has a similar serum biochemical profile, calculation of the fractional excretion of calcium (FeCa; calculated using a 24-hour urine sample collected off diuretics) has traditionally been used. Values below 1% are consistent with FHH, but overlap of FeCa values in FHH and PHPT occurs.As FeCa values can also be low in patients with PHPT who have coexisting vitamin D deficiency, the diagnosis of FHH should not be made until vitamin D stores are replete. FeCa can also be misleading in patients with impaired renal function (due to advanced age or renal disease). In these patients, obtaining past serum calcium levels (which should be consistently elevated) and family history of hypercalcaemia is helpful.
Ultimately, if suspicion of FHH is high, mutational analysis of CASR for FHH1, as well as mutational analysis of GNA11 and AP2S1 for the diagnosis of FHH2 and FHH3, respectively, can be performed differentiation of PHPT from FHH is important, as surgical intervention is not indicated or curative in FHH.
PHPT can be distinguished from secondary and tertiary hyperparathyroidism by its different biochemical profile

Secondary hyperparathyroidism is associated with an appropriate elevation in PTH in response to a hypocalcaemic stimulus and either a frankly low or normal serum calcium level. Most commonly, secondary hyperparathyroidism is due to vitamin D deficiency, malabsorption, kidney disease or hypercalciuria.
Tertiary hyperparathyroidism describes a condition in which prolonged, severe secondary hyperparathyroidism (as in end-stage renal disease) evolves into a hypercalcaemic state due to autonomous functioning of hyperplastic parathyroid glands. Although this effect can be observed in patients on dialysis, it can also occur after renal transplant. Tertiary hyperparathyroidism is typically obvious
from the history of the patient.
Clinical manifestations and complications
Classical PHPT
Symptomatic, multi-system disorder characterized by skeletal, renal, gastrointestinal, neurological and psychiatric manifestations classical PHPT is uncommon today in the USA, Western Europe and Turkey.
Marked hypercalcaemia (11.5–16.8mg/dl), osteitis fibrosa cystica, a skeletal condition characterized clinically by bone pain and fractures (particularly vertebral) and radiographically by demineralization, fibrosis, brown tumours and bone cysts. Nephrolithiasis, nephrocalcinosis, polyuria and polydipsia as well as renal impairment.
Asymptomatic PHPT
Today, the vast majority (>80%) of patients with PHPT in the USA and Western Europe are ‘asymptomatic’,a term used to describe those lacking the skeletal and renal manifestations described in classical PHPT.
Biochemical profile
Most patients with PHPT are incidentally discovered when routine laboratory work reveals hypercalcaemia. (often within 1mg/dl of the upper limit of normal) PTH levels are typically within two times the upper limit of normal. Serum phosphate is often in the lower half of the normal range and less frequently frankly low due to the phosphaturic effects of PTH. Alkaline phosphatase levels can be elevated, a reflection of increased bone resorption and compensatory formation, but most often remain within the normal range.
The storage form of vitamin D, 25-hydroxyvitamin D (25OHD), is often in the insufficient (20–29ng/ml) or deficient range (<20 ng/ml), whereas activated vitamin D (1,25-dihydroxyvitamin D; 1,25(OH)2D) is near the upper end of normal and sometimes frankly elevated. Potential pathophysiological mechanisms for vitamin D deficiency in PHPT include the following: PTH enhances the conversion of 25OHD to 1,25(OH)2D by inducing the renal 1α-hydroxylase enzyme45, the half-life of 25OHD might also be shortened due to enhanced hepatic in
activation46, and chronic vitamin D deficiency could result in parathyroid hyperplasia and autonomous adenomatous change.
Skeletal manifestations
Dual-energy X-ray absorptiometry (DXA) demonstrates preferential loss of BMD at cortical sites such as the distal one-third of the forearm cancellous sites such as the lumbar spine are relatively spared reflects the catabolic versus anabolic effects of PTH on different skeletal compartments.
Estimates of the prevalence of osteoporosis in PHPT have varied in recent studies (39–62.9%). Epidemiological data, however, suggest an increased risk of both vertebral and peripheral fractures. The paradox of increased vertebral fracture risk despite preserved lumbar spine BMD in PHPT has remained unclear until recently.
Renal manifestations
Hypercalciuria and nephrolithiasis. Risk factors for nephrolithiasis include younger age and male sex, whereas degree of hypercalcaemia and hypercalciuria, PTH levels and other urinary factors have shown less consistent associations. The prevalence
of renal dysfunction (estimated glomerular filtration rate (eGFR) <60ml/min) is low, with recent studies suggesting rates of 15–17%
Neuropsychological features
Calcium has a key role in regulating the release of neurotransmitters at synapses, and hypercalcaemia could interfere with that process. Vascular effects of PTH could also affect cognition and mood by altering cerebrovascular function. A number of studies suggest, however, that even ‘mild PHPT’ (serum calcium <12mg/dl) is associated with nonspecific symptoms such as depression, anxiety, fatigue, decreased quality of life (QoL), sleep disturbance and cognitive dysfunction. Many, but not all, observational studies have indicated these features improve after parathyroidectomy.
Cardiovascular manifestations
Hypertension has long been associated with PHPT but is not reversible with parathyroidectomy. Recent studies have investigated subclinical cardiovascular findings in PHPT, including asymptomatic coronary artery disease (CAD), valve calcification, left ventricular hypertrophy (LVH), carotid disease and vascular stiffness.
Normocalcaemic PHPT
Biochemical profile
NPHPT as a phenotype of PHPT. Prevalence of 0.4–3.1% in community-based cohorts. Although calcium levels are normal in this entity, PTH levels are similar or slightly lower than in the hypercalcaemic form of PHPT. By definition, ionized calcium, vitamin D levels, urinary calcium excretion and renal function must be normal to distinguish this entity from secondary hyperparathyroidism. NPHPT can precede the development of typical hypercalcaemic PHPT, but patient series have suggested only 0.6–19% of patients go on to develop hypercalcaemia.
Evaluation and management
Surgery
Parathyroidectomy remains the only cure for PHPT and is recommended in all symptomatic patients.
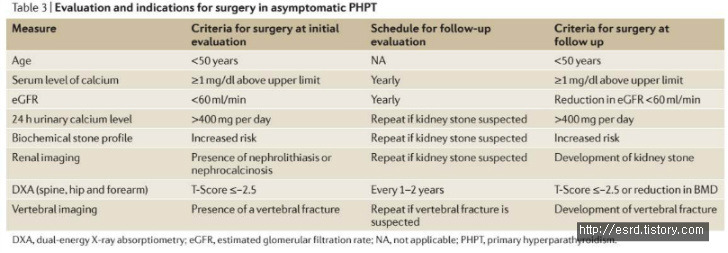
The Fourth International Workshop for the Management of Asymptomatic PHPT recommended surgical intervention in patients with asymptomatic PHPT in whom evidence of subclinical end-organ (skeletal or renal) effects or risk of disease progression exists.
Preoperative localization is necessary if a minimally invasive parathyroidectomy (MIP) is contemplated. Cervical ultrasonography can localize parathyroid disease and assess for concomitant thyroid pathology. Technetium (99mTc) sestamibi is the dominant radioisotope in parathyroid scintigraphy. Combined ultrasonography and sestamibi imaging increases localization accuracy and improves sensitivity. Sestamibi protocols have low sensitivity in multi-gland disease.
Preoperative fine-needle aspiration biopsy (FNAB) preoperatively is generally not recommended in PHPT and is absolutely contraindicated if parathyroid cancer is a diagnostic consideration, as FNAB can seed the operative site, leading to spread of disease.
Non-surgical monitoring and management
Long-term observational studies indicate that biochemistries and BMD remain stable for many years in those followed non-operatively. However, 15-year data suggest that BMD starts to decline at cortical sites after 10 years of observation, and almost 40% of patients developed one or more indications for parathyroidectomy over 15 years of follow-up. Regular monitoring of biochemistries and of BMD with DXA is recommended repeat imaging of the spine and kidney is advised when vertebral fractures or nephrolithiasis is suspected.
All patients who are observed should be advised to stay adequately hydrated and not to restrict dietary calcium intake. Liberal dietary calcium intake does not worsen hypercalcaemia. Conversely, restriction could exacerbate hyperparathyroidism. Recent guidelines recommend restoring vitamin D to levels of 21–30 ng/ml with conservative doses of vitamin D (600–1000 IU daily) on the basis of data showing that vitamin D repletion lowers PTH levels. Higher levels of vitamin D might be beneficial.
Ideal medical therapy of PHPT would provide the equivalent to a medical parathyroidectomy. Such an agent would normalize serum calcium and PTH levels as well as urinary calcium excretion, increase BMD and lower fracture risk, and reduce the risk of kidney stones. Unfortunately, no currently available single drug meets all these criteria.
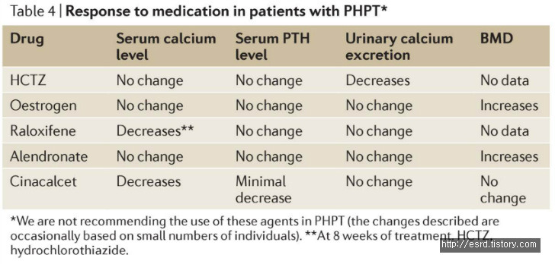
Hydrochlorothiazide
Hydrochlorothiazide (12.5–50mg daily for 3.1 years on average) was associated with a decrease in urinary calcium excretion but no change in serum levels of calcium thiazides could be considered in those who refuse surgery or are poor surgical candidates but at high risk of nephrolithiasis, as long as serum levels of calcium are monitored regularly.
Oestrogen and selective oestrogen receptor modulators
An RCT of conjugated oestrogen (0.625mg daily plus medroxyprogesterone at 5 mg daily) versus placebo indicated that hormone-replacement therapy effectively increases BMD at all skeletal sites in patients with PHPT, with the greatest increases at the lumbar spine. This RCT, however, did not confirm the calcium-lowering effect of earlier uncontrolled studies.
Bisphosphonates and denosumab
Several small RCTs have indicated that alendronate increases BMD at the lumbar spine and hip in patients with PHPT, but most studies suggest there is no change in serum biochemistries. No data exist regarding fracture risk reduction with alendronate, and no BMD data are available in PHPT. No published data are available regarding the use of denosumab in PHPT.
Cinacalcet
Cinacalcet is a type 2 calcimimetic that binds to the CASR and increases its sensitivity. Cinacalcet effectively reduces serum levels of calcium in patients with PHPT. Cinacalcet was approved for PHPT by the European Medicines Agency in 2008 and by the FDA in 2011 for the treatment of severe hypercalcaemia in patients with PHPT who are unable to undergo parathyroidectomy. Unfortunately, neither BMD nor urinary calcium excretion improves with cinacalcet treatment. Cinacalcet is also frequently associated with headache, nausea and vomiting.
** 인상적인 내용을 요약
- 1. Primary hyperparathyroidism 은 parathyroid cell mass 도 증가하고, Calcium sensing receptor 의 sensitivity 도 감소하여 PTH의 분비를 억제하기위해 많은 calcium 이 필요하다는 것.
- 2. 최근 검진의 증가로 Classical PHPT -> Asymptomatic PHPT -> Normocalcemic PHPT 등의 용어가 생겨나고 있음. 여기서 asymptomatic PHPT 의 정의는 hypercalcemic 하고 PTH는 증가되어 있으나 bone 이나 renal 의 변화는 (아직) 없다는 것.
- 3. PHPT 의 경우 BMD 를 해보면 주로 cortical bone의 BMD loss 가 일어나나 lumbar spine 은 Spared 됨.
- 4. 약물 치료하며 경과를 볼 때는 calcium 과 vitamin D 를 안줘야할 것 같은데, 주는게 낫다는 점.
- 5. 그 외 Hydrochlorothiazide, Estrogen, Raloxifene, Alendronate, Cinacalcet 등을 써볼 수 있겠다.
- 특히 calcium 을 낮추고 싶을 때는 cinacalcet. (물론 calcium 이 엄청 높으면 수술을 해야지.)